 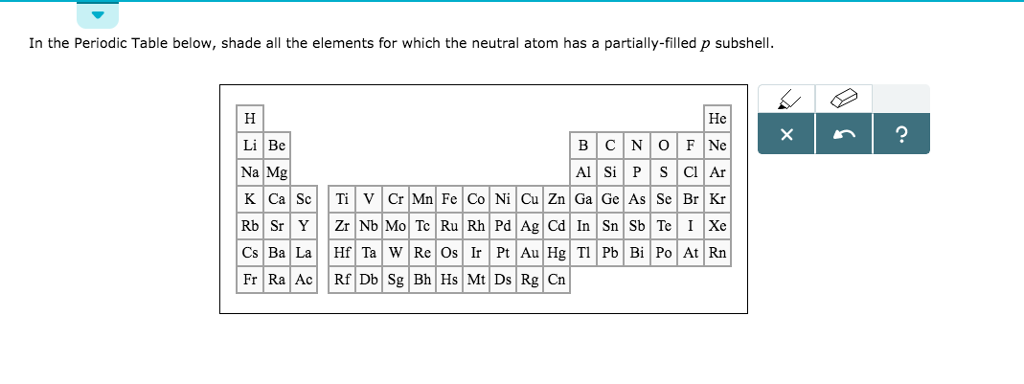
The decrease in size is regular in ions but not so regular in atoms. Inner transition elements: As we move along the lanthanide series, there is a decrease in atomic as well as ionic radius. Impact of 3d-transition metal T Sc, Ti, V, Cr, Mn, Fe, Co on praseodymium perovskites PrTO3: standard spin-polarized GGA and GGA+U investigations.The distance between two cores will give the measurement of a particle however you need the span which is a large portion of the width. 1.44 1.32 1.22 1.18 1.17 1.17 1.16 1.15 1.17 1.25. At the point when two iotas of a similar component are covalently reinforced, the span of every particle will be a large portion of the distance between the two cores since they similarly draw in the electrons. The elements of 3d transition series are given as: Sc Ti V Cr Mn Fe Co Ni Cu Zn Answer the following: (i) Write the elements which is not regraded as. (iv) is a strong oxidizing agent, as it readily accepts an. (iii) Copper (Cu) (Atomic number 29) can show oxidation state is +1. We have to know that at the point when a covalent bond is available between two iotas, the covalent range can be resolved. Answers (1) (i) Zinc (Zn) is not regarded as transition element since it does not contain partially filled d-orbital in either ground state or stable +2 oxidation state. The conditions on climate, test, and state lead to a variety of definitions. 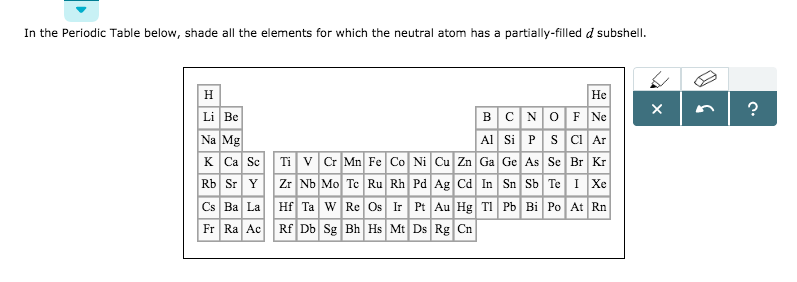
Commonly, on account of the trouble to disconnect iotas to quantify their radii independently, nuclear range is estimated in a reinforced state anyway hypothetical computations are obviously easier while thinking about molecules in separation. The theoretical study of Au n M + clusters (MSc, Ti, V, Cr, Mn, Fe, Au n 9) discovered that the positive charge of the cationic Au n M + clusters, which is localized mainly in the impurity, and the magnetic and geometrical configurations are strongly correlated with cluster sizes. Four generally utilized meanings of nuclear span are: Van der Waals sweep, ionic range, metallic span and covalent sweep. Since the limit is definitely not a clear cut actual element, there are different non-identical meanings of nuclear span. We must have to remember that the atomic radii of a synthetic component are a proportion of the size of its particles, normally the mean or regular separation from the focal point of the core to the limit of the encompassing shells of electrons. As you get across or down the intermittent table, patterns arise that help clarify how nuclear radii change. In less complex terms, it tends to be characterized as something like the range of a circle, where the focal point of the circle is the core and the external edge of the circle is the peripheral orbital of electrons. 
Hint: We know that Atomic radius is by and large expressed similar to the absolute separation from a molecule's core to the furthest orbital of electrons. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
